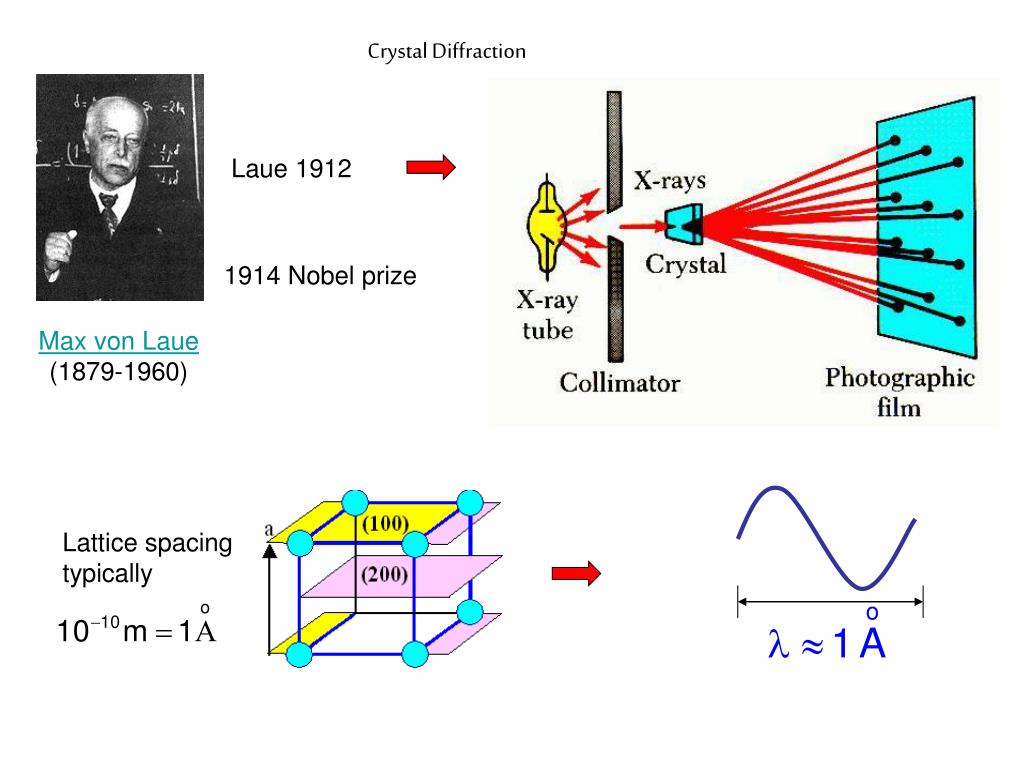
Serial femtosecond crystallography (SFX) was developed at XFELs and more than a hundred structures have been determined using the approach, as revealed by the number of depositions to the protein databank 3. Instead of rotating a large single crystal in the X-ray beam while acquiring a series of consecutive diffraction patterns, in serial crystallography (SX) datasets are collected by taking individual snapshots from hundreds to hundreds of thousands of microcrystals and then merging the diffraction data into a single and complete three-dimensional dataset. Recent advances in highly brilliant X-ray sources, fast frame-rate detectors and novel sample delivery techniques have changed the way crystallographic data can be collected at both X-ray free electron lasers (XFELs) and third generation synchrotron sources 1, 2. The high quality of the diffraction data highlights the potential of this method for studying irreversible reactions at sub-microsecond timescales using high-brightness X-ray facilities. 
In contrast to experiments with monochromatic radiation, data from only 50 crystals were required to obtain complete datasets. Here we report the structure determination of two different protein samples by merging pink-beam diffraction patterns from many crystals, each collected with a single 100 ps X-ray pulse exposure per crystal using a setup optimized for very low scattering background. The polychromatic, “pink”, beam provides a more than two orders of magnitude increased photon flux and hence allows accessing much shorter timescales in diffraction experiments at synchrotron sources. The time resolution for serial synchrotron crystallography experiments has been limited to millisecond timescales with monochromatic beams. Contrary to traditional protein crystallography, convergent beam systems using capillary optics can be used for screening very small protein crystals, and micro diffraction measurements can be made using a very low-powered source.Serial X-ray crystallography allows macromolecular structure determination at both X-ray free electron lasers (XFELs) and, more recently, synchrotron sources. The diffracted-beam intensity obtained with a polycapillary monolithic optic with a slightly convergent beam and a 50 W microfocus source was equal to or greater than that from a 5 kW rotating anode source equipped with the most advanced confocal optics. Single crystal convergent beam X-ray diffraction using a polycapillary focusing optic Single crystal parallel beam X-ray diffraction using a polycapillary collimating optic Even smaller unit cell sizes can be measured with micro X-ray diffraction using focusing optics for more strongly convergent beams. 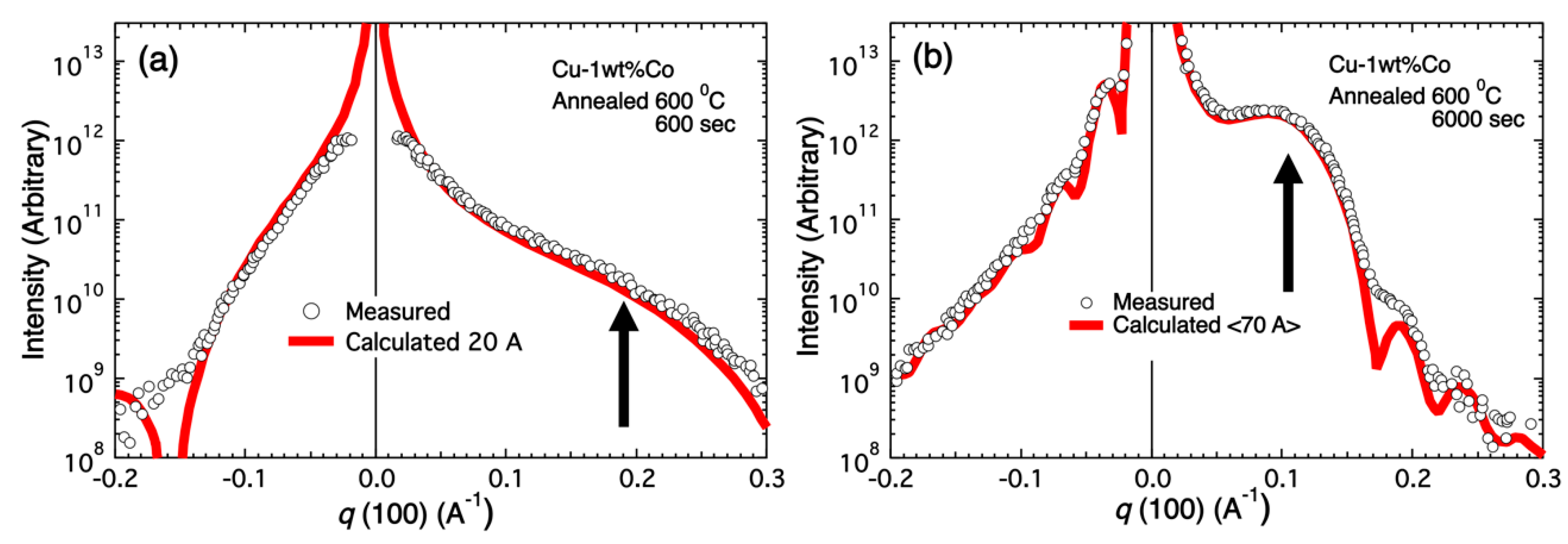
The unit cell size that can be successfully analyzed with collimating or slightly focusing optics is < 200?÷. Polycapillary optics can be used in conjunction with low-power X-ray sources to achieve X-ray densities in small beam spots, resulting in diffracted-beam intensities equal or greater than that achieved using rotating anode sources equipped with the most advanced confocal optics. 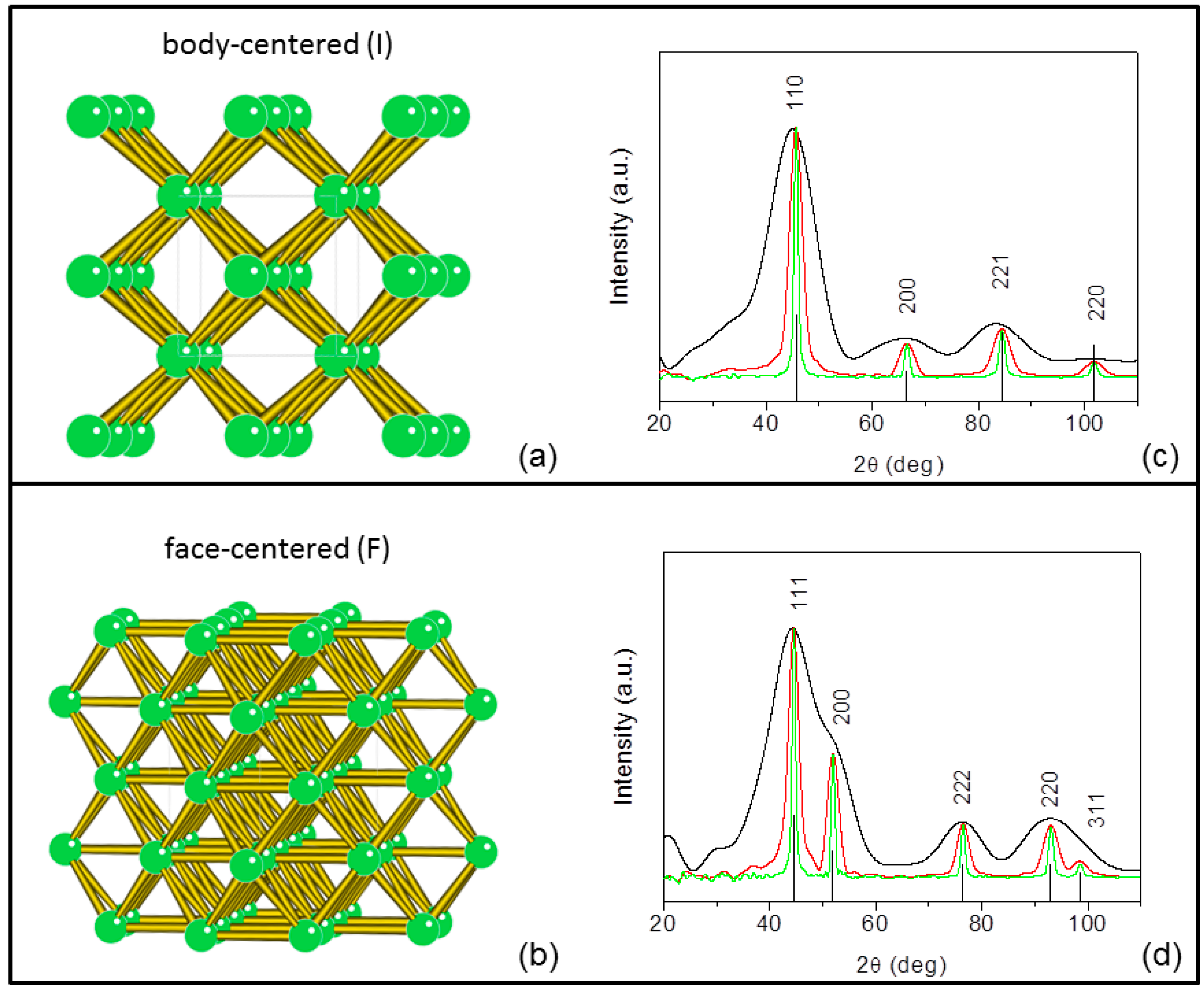
Contrary to traditional protein crystallography, convergent beam systems using capillary optics can be used for screening very small protein crystals, and micro diffraction measurements can be made using a very low-powered source. Single crystal diffraction or protein crystallography measurements can be enhanced by using either polycapillary collimating or polycapillary focusing optics. Since protein and other biomacromolecular crystals are very weak diffractors, it is often desirable to obtain increased flux on the crystal with conventional laboratory X-ray sources.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
